From the Editor
State-of-the-art care for acute coronary syndrome includes oxygen and clot-busting drugs. Should it also include a depression screen and an antidepressant if necessary?
In this week’s Reading, we look at a new JAMA paper showing a response to escitalopram for patients post-ACS (Acute Coronary Syndrome) with depression. Though work has been done in this area before, this paper is an important contribution: it’s well designed, and offers a long follow-up period. Chonnam National University Medical School’s Jae-Min Kim and his co-authors conclude: “In this median 8.1-year follow-up of a randomized 24-week clinical trial of treatment for depression in patients with recent ACS, MACE [major adverse cardiac event] incidence was significantly lower in patients receiving escitalopram than those receiving placebo.”
We consider the paper and its implications.
 Good EKG, good antidepressant?
Good EKG, good antidepressant?
Please note that there will be no Readings for the next two weeks.
DG
Meds and Outcomes
“Effect of Escitalopram vs Placebo Treatment for Depression on Long-term Cardiac Outcomes in Patients With Acute Coronary Syndrome: A Randomized Clinical Trial”
Jae-Min Kim, Robert Stewart, Yong-Seong Lee, Hee-Joon Lee, Min Chul Kim, Ju-Wan Kim, Hee-Ju Kang, Kyung-Yeol Bae, Sung-Wan Kim, Il-Seon Shin, Young Joon Hong, Ju Han Kim, Youngkeun Ahn, Myung Ho Jeong, Jin-Sang Moon
JAMA, 24/31 July 2018
https://jamanetwork.com/journals/jama/article-abstract/2688569
Depression is frequently comorbid with acute coronary syndrome (ACS; including myocardial infarction [MI] and unstable angina) and associated with poor outcomes including increased mortality and nonfatal events. A clinically important question is whether antidepressant treatment mitigates these adverse effects. In randomized clinical trials of antidepressants in ACS, mostly evaluating selective serotonin reuptake inhibitors, improvement in depressive symptoms has been demonstrated repeatedly. However, effects on cardiac outcomes have not generally been found within the treatment period.
Considering longer-term cardiac outcomes, in an 18-month follow-up of the Myocardial Infarction and Depression–Intervention Trial (MIND-IT), no difference was found in cardiac outcomes between 209 patients receiving antidepressants (8 weeks of mirtazapine and/or subsequent open-label citalopram) and 122 patients receiving usual care. In the 6.7-year follow-up of the Sertraline Antidepressant Heart Attack Randomized Trial (SADHART), there was also no difference in mortality between 184 patients receiving 6 months of sertraline and 177 receiving placebo. Furthermore, the Enhancing Recovery in Coronary Heart Disease Patients (ENRICHD) trial found no difference in cardiac outcomes over a 29-month follow-up between the intervention (9 months of cognitive behavioral therapy with or without 12 months of antidepressants) and usual care among 2481 patients with depression following MI. However, in a subanalysis, 301 patients receiving selective serotonin reuptake inhibitors showed better cardiac outcomes than 1388 not receiving them. Conversely, an observational study reported that 58 patients who received selective serotonin reuptake inhibitors had worse outcomes than 354 who did not receive them over a 3.6-year follow-up.
Associations of antidepressant treatment with long-term cardiac outcomes in depression following ACS have therefore been inconclusive. Previous research has been limited by short follow-up periods, heterogeneous antidepressant regimes and samples, and limited evaluation of cardiac outcomes. This study aimed to investigate whether the effect of escitalopram vs placebo for treating acute-phase depression in patients with recent ACS resulted in benefits in longer-term cardiac outcomes.”
 Jae-Min Kim
Jae-Min Kim
So begins a paper by Kim et al.
Here’s what they did:
- They worked with South Korean inpatients who had had an ACS in the previous two weeks, and agreed to depression screening – which included the Beck Depression Inventory and (with scores over 10) a clinical evaluation by a study psychiatrist.
- Those with depression were randomized to a 24-week, double-blind, randomized clinical trial of escitalopram vs. placebo. The escitalopram dosing was 10 mg daily initially, but may have been changed, from 5 mg to 20 mg daily, depending on clinician judgment and tolerability.
- “The primary outcome was remission of depressive symptoms, defined by a Hamilton Depression Rating Scale (HAM-D) score of 7 or lower at the trial end.”
- “The primary end point was MACE, which was a composite of all-cause mortality, MI, and PCI [percutaneous coronary intervention].”
- Various statistical analyses were performed.
Here’s what they found:
- “Of 4809 patients with ACS screened, 1152 were recruited to undergo depression screening with the BDI and MINI, through which 446 participants were identified as having a baselineDSM-IV diagnosis of major (n = 202) or minor (n = 246) depressive disorder.”
- 300 were then included in the trial. They were randomized to the escitalopram group (149) or placebo (151).
- Demographically: patients tended to be around 60, and higher in their BDI score (16 and 17, for the escitalopram and placebo groups, respectively). Men were more represented than women.
- “All participants were followed up for 5 to 11 years until death or June 2017 (median follow-up, 8.1 [interquartile range, 7.1-9.0] years…).”
- “Before the end of the 24-week trial, 12 patients experienced MACE… A significant difference was found: composite MACE incidence was 40.9% (61/149) in the escitalopram group and 53.6% (81/151) in the placebo group (hazard ratio [HR], 0.69)…”
- “Remission was achieved in 57 (52.3%) of 108 escitalopram patients and in 38 (34.9%) of 109 placebo participants who completed the 24-week trial.”
- “With respect to the association with treatment group, those randomized to escitalopram had significantly lower hazards of composite MACE, MI, and PCI compared with those randomized to placebo.” See graph below.
- For a visual summary of the full study, see figure below.
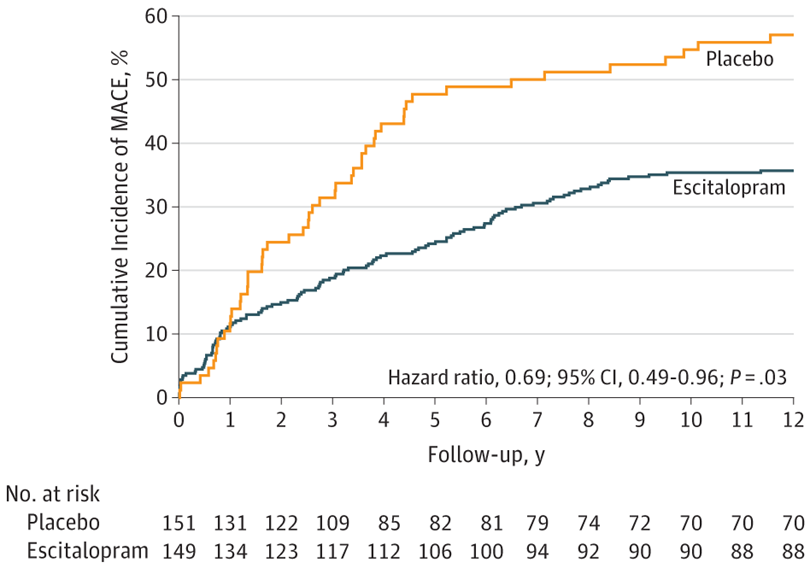

A few thoughts:
- This is a good study.
- For years, people have debated whether antidepressants are effective for depression. Major work – including by Cipriani et al. – suggests that they are. This paper adds to the literature, showing that for those with medical illness, the difference between using antidepressants and not using antidepressants may be life and death. After six months of treatment, patients taking the antidepressant were offered protection against adverse events years (8.1) out. I’ll note that the NNT (number needed to treat) was between 5 and 6 – not incredible, but enough to make a difference in outcomes.
- How to reconcile these robust results with past trials? The authors forward a few observations, including the ethnicity of the patients and the longer treatment in this study than in one of the past studies. Also: “the levels of depressive symptoms in this study’s participants were less severe (mean baseline HAM-D score, 15.9) than in MIND-IT or SADHART (mean baseline HAM-D scores, 18.1 and 19.6, respectively).Of potential relevance, previous research has indicated that even minor depressive symptoms may have significant negative effects on cardiac prognosis…”
- For all of the press about withdrawal syndrome, it would seem that the vast majority of patients were able to titrate out their antidepressants.
- On Twitter, psychiatrist Peter Kramer (who wrote the book Listening to Prozac) speculates that the NNT for escitalopram and cardiac disease with depression may be better than statins with this subpopulation. Interesting.
- Putting this in a larger perspective: if we undertreat depression in our society (consider the work of Patten et al. showing that only half of Canadians with depression receive minimally adequate care), how are we doing with people post-MI?
- The Reading of the Week will return in a fortnight, post-Maritime lobster. If you have any suggestions for future Readings, please send an email.
Reading of the Week. Every week I pick articles and papers from the world of Psychiatry.
Recent Comments