From the Editor
Is this the breakthrough we have been waiting for?
This week, we look at the new study from The New England Journal of Medicine considering psilocybin (a naturally occurring psychedelic compound found in some mushrooms) and escitalopram for depression. The paper, written by Robin Carhart-Harris (of Imperial College) and his co-authors, has received much attention. One online news source quipped: “Tripping may be as effective as your antidepressants.”
In this study, psilocybin was compared to the SSRI with a double-blind, randomized, controlled trial. Carhart-Harris et al. find: “On the basis of the change in depression scores on the QIDS-SR-16 at week 6, this trial did not show a significant difference in antidepressant effects between psilocybin and escitalopram in a selected group of patients.”

We consider the paper and the editorial that runs with it by Dr. Jeffrey A. Lieberman (of Columbia University). We also ask Dr. Ishrat Husain (of the University of Toronto) for his thoughts. Finally, we mention other cutting-edge treatments.
DG
“Trial of Psilocybin versus Escitalopram for Depression”
Robin Carhart-Harris, Bruna Giribaldi, Rosalind Watts, et al.
The New England Journal of Medicine, 15 April 2021

Major depressive disorder affects approximately 10% of the general population in the United Kingdom, impairs patients’ lives, and is costly to society. Selective serotonin-reuptake inhibitors are first-line treatments for major depressive disorder; however, these drugs take several weeks to work and, in some patients, do not induce a response. Escitalopram, a selective serotonin-reuptake inhibitor, is representative of the currently used antidepressants in terms of safety and efficacy.
The psychedelic compound psilocybin is the phosphorylated ester of its metabolite, psilocin (4-OH-N,N-dimethyltryptamine). Psilocybin and psilocin occur naturally in the psychoactive psilocybe genus of mushrooms. As with other traditional psychedelic substances, the main effects of psilocin occur through serotonin 5-hydroxytryptamine type 2A (5-HT2A) receptor agonism, which is part of a pathway implicated in depression. Psilocybin showed promise as an adjunct to psychotherapy for mood disorders and addiction in the mid-20th century.
One open-label trial and four randomized, controlled clinical trials of psilocybin for depression and anxiety have been conducted. Reductions in depressive symptoms after the administration of one or two doses of psilocybin were observed in trials across several patient populations, including a small open-label trial involving patients with treatment-resistant depression, the results of which informed the current trial. We performed a phase 2, double-blind, randomized, controlled trial involving patients with long-standing, moderate-to-severe major depressive disorder to compare psilocybin with escitalopram over a 6-week period.
So opens a paper by Carhart-Harris et al.
Here’s what they did:
- They performed “a phase 2, double-blind, randomized, controlled trial involving patients with long-standing, moderate-to-severe major depressive disorder…”
- Psilocybin was compared to escitalopram over a 6-week period.
- “Patients were assigned in a 1:1 ratio to receive two separate doses of 25 mg of psilocybin 3 weeks apart plus 6 weeks of daily placebo (psilocybin group) or two separate doses of 1 mg of psilocybin 3 weeks apart plus 6 weeks of daily oral escitalopram (escitalopram group); all the patients received psychological support.”
- The primary outcome was the change from baseline in the score on the 16-item Quick Inventory of Depressive Symptomatology–Self-Report.
Here’s what they found:
- 59 people were enrolled, with 30 in the psilocybin group and 29 in the escitalopram group.
- Demographics: there were more men than women (63% in the psilocybin group and 69% in the escitalopram group); people tended to be employed (70% and 72%); they weren’t regular users of psilocybin (72% and 74%).
- The mean scores on the QIDS-SR-16 at baseline were 14.5 in the psilocybin group and 16.4 in the escitalopram group.
- The mean changes in the scores from baseline to week 6 were −8.0 in the psilocybin group and −6.0 in the escitalopram group, for a between-group difference of 2.0 points.
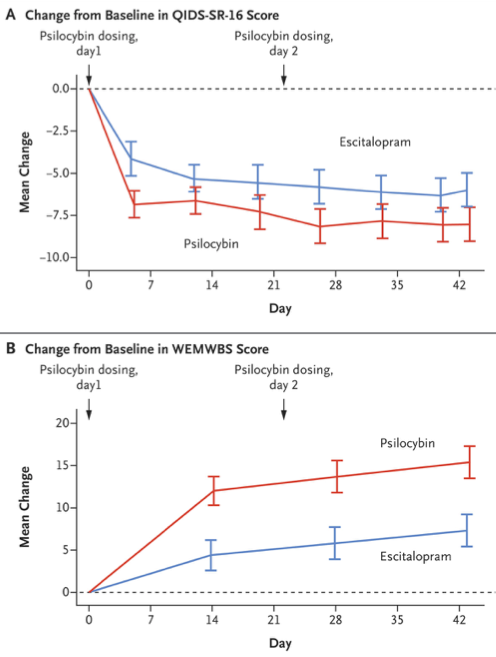
A few thoughts:
- This is a good paper.
- About the results – the core finding is that psilocybin matched escitalopram (remember, with the study design, a negative result is actually a positive). The visual abstract does a terrific job of summarizing the paper:
- There was a difference with the secondary outcomes. But, to emphasize: those are secondary outcomes.
To highlight from the paper: “A QIDS-SR-16 response at 6 weeks occurred in 21 patients (70%) in the psilocybin group and in 14 patients (48%) in the escitalopram group… QIDS-SR-16 remission at week 6 occurred in 17 patients (57%) in the psilocybin group and in 8 patients (28%) in the escitalopram group…”
- Like all studies, there are limitations. The authors note several including “the brief duration of escitalopram treatment, because this drug has a delayed therapeutic action on depression. Had the course of escitalopram been extended, it is possible that better efficacy would have been observed among the patients in the escitalopram group.”
- How to understand the paper? I asked Dr. Ishrat Husain, who has published on new treatments for mood disorders, to weigh in.

I think it is fantastic that The New England Journal of Medicine is publishing original mental health research and it is a huge achievement by the authors to have their study published by the premier medical journal. The authors should be congratulated for completing a randomized controlled trial of a psychedelic compound, psilocybin, that can be difficult to blind due to the profound psychoactive effects of the drug. That being said, this is effectively a phase II/pilot study that included only 59 participants, and was powered on findings from open-label studies, which notoriously inflate effect sizes. Hence the power calculation for the primary outcome was unlikely to be reliable. Although the authors report that psilocybin was better than escitalopram on all secondary outcomes, technically secondary outcomes should not be analyzed and reported for preliminary/pilot trials as the study is not powered to assess these measures. I think this is particularly problematic for this study as many media outlets have been using the secondary findings to promote psilocybin as being better than antidepressants!
Although psilocybin is not a panacea, the study provides the strongest evidence for its potential antidepressant effect. The safety and tolerability data is also encouraging and shows that with careful, controlled administration, these treatments can be given safely. However, we need much more data from well designed and larger RCTs before these treatments can be translated in to clinical practice – we still don’t know which patients would be most appropriate for psychedelic treatments nor do we know how long their effects last. I hope this study encourages further research and importantly influences funders to support more trials to allow for further investigation of novel treatments like psychedelics, which are urgently needed in mental health.
- The paper runs with an editorial by Dr. Jeffrey A. Lieberman.
He opens by noting that there is nothing new under the sun. Drawing on the history of psychiatry, he writes: “Excitement over psychedelic drugs led to extravagant claims about their vast potential to expand human consciousness, elucidate the psychological architecture of the brain, and treat mental disorders. By the mid-1960s, LSD had been prescribed to approximately 40,000 patients in the United States and spawned over 1000 scientific papers, dozens of books, and cover stories in Time and LIFE magazines. Meanwhile, recreational use of these drugs, encouraged by countercultural icons Timothy Leary and Ken Kesey, spread. Appeals to ‘tune in, turn on, and dropout’ propelled unsupervised use to leap-frog medical research.”
Turning his attention to this study, he wonders about the mechanism of action. “Although this trial is an evidentiary milestone in the development of psychedelic drugs, it also reveals major knowledge gaps. It is unknown how these drugs produce their mind-altering effects. Psychedelics are partial agonists at the 5-hydroxytryptamine type 2A (5-HT2A) receptor, but so are drugs such as lisuride that do not produce subjective effects. Possible explanations of the different effects of psychedelics include functional selective biophysical engagement of the 5-HT2A receptor and activation of alternative intracellular singling pathways such as β-arrestinand the ability to alter neural circuitry by stimulating proliferation of dendritic spines and synapse formation. The latter explanation is challenged by the temporal dissociation of the immediate subjective experience and the subsequent neurobiologic alterations induced by psychedelic drugs. Another hypothesis is that psychedelics inactivate the prediction-error minimization function of the brain, thereby weakening the mental mechanisms that maintain one’s sense of self.”
The editorial can be found here:
https://www.nejm.org/doi/full/10.1056/NEJMe2102835
- A couple of years ago, Carhart-Harris suggested in an interview that psilocybin may one day replace antidepressants; last week, in The Guardian, he wrote that we may be “on the verge of a paradigm shift in mental healthcare linked to an improved understanding of the origins of depression, and how we can most effectively treat it.” This study is interesting but doesn’t quite support that sweeping view.
- Interested in reading more about novel treatments? Here are a few papers worth looking at:
Dr. Adriana Feder (of Mount Sinai) and her co-authors have recently published an RCT on ketamine for PTSD in The American Journal of Psychiatry.
https://ajp.psychiatryonline.org/doi/10.1176/appi.ajp.2020.20050596
The paper runs with an editorial by Drs. Murray B. Stein (of the University of California San Diego) and Naomi M. Simon (of New York University) with the clever title: “Ketamine for PTSD: Well, Isn’t That Special.”
https://ajp.psychiatryonline.org/doi/10.1176/appi.ajp.2020.20121677
Dr. Husain and his co-authors explore minocycline and celecoxib as adjuctive treatments for bipolar depression in Lancet Psychiatry.
https://www.thelancet.com/journals/lanpsy/article/PIIS2215-0366(20)30138-3/fulltext
Tom K. J. Craig (of King’s College) and his co-authors do a study using AVATAR therapy for auditory hallucinations in Lancet Psychiatry.
https://www.thelancet.com/journals/lanpsy/article/PIIS2215-0366(17)30427-3/fulltext
That paper was considered in a past Reading:
And the Carhart-Harris paper can be found here:
https://www.nejm.org/doi/full/10.1056/NEJMoa2032994
Reading of the Week. Every week I pick articles and papers from the world of Psychiatry.
Recent Comments