From the Editor
The tone of our voice when discussing a treatment. The way we emphasize its benefits. Mention of our hopefulness. As clinicians, we are aware that small things can improve patient outcomes. To speak more technically: we harness placebo effects.
In an impressive new Lancet Psychiatry review, Dr. Matthew J. Burke (of the University of Toronto) and his co-authors examine placebo effects with an eye to the implications for clinical practice. They discuss imaging and physiology; they also mention depression and offer suggestions (including, yes, harnessing placebo effects). “The thoughtful application of placebo and nocebo effects has promise in enhancing patient care and treatment outcomes, but more research is needed to validate specific approaches.” We discuss the paper and its implications.

What is the survival rate after an opioid overdose? In a new research letter just published in JAMA, Dr. Robert A. Kleinman (of the University of Toronto) and his co-authors attempt to answer that question. Though they walk on a well-travelled path, they drew on data in this, the era of fentanyl. “Mortality was higher than estimates from previous research conducted prior to the widespread availability of fentanyl (5.3%-5.5%).”
And in the third selection from The Globe and Mail, Dr. Lester Liao (of McGill University) and his co-authors write about mental health campaigns. They note the unintended consequences, including overdiagnosis of disorders like ADHD and autism. “We started out with the greatest of intentions. Unintended consequences have arisen. That’s okay. We learn along the way. Now it’s time to change course.”
There will be no Reading next week.
DG
Selection 1: “Harnessing placebo effects and mitigating nocebo effects: implications for clinical practice in psychiatry and medicine”
Matthew J. Burke, Dasha A. Sandra, Marta Peciña, et al.
The Lancet Psychiatry, May 2026
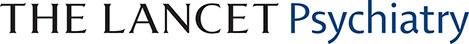
In psychiatry and medicine, there is a long history of framing placebo effects primarily as nuisance factors and focusing on how they should be minimised in clinical trials. However, a new view on placebo effects has emerged with advances in understanding their complex neurobiology and observations of unexpectedly large placebo responses in recent psychiatric trials. In particular, novel therapeutic device trials for depression have shown placebo-group remission rates nearing 50%. Instead of considering these studies as a failure and moving on, questions should be raised on how such responses are possible and how these effects can be harnessed for the benefit of patients.
So begins a paper by Burke et al.
I. Introduction
“Placebo effects can be defined as therapeutic benefits derived from the context surrounding administration of a treatment rather than the treatment itself. These effects involve a bidirectional interplay between numerous external factors (eg, environmental cues, patient–clinician interactions, and societal considerations) and internal factors (eg, beliefs, emotions, previous experience, interpersonal scripts, and cognitive schemas)… Evolving neuropsychological models emphasise the central role of expectancies in generating placebo effects, which can encompass both conscious (expectations) and unconscious processes.”
II. Imaging and Physiology
“Over the past 20 years, neuroimaging and neurophysiological studies have shown that placebo effects are capable of meaningfully modulating brain networks and neurotransmitter systems. This research has been supported by studies demonstrating the ability to block placebo effects pharmacologically, in a similar way to blocking the action of drugs (eg, reduction of placebo effects with the opioid antagonist naloxone), observed dose–response relationships associated with experimental manipulation of placebo effects, and the striking loss of treatment efficacy when contextual cues and expectancies are removed from a therapeutic interaction (eg, open-hidden paradigms). Placebo effects have also been increasingly studied in animal models—one study that used various advanced cellular and molecular techniques reported placebo analgesia in mice mediated by a pathway from the rostral anterior cingulate cortex to the brainstem and cerebellum. Many of the placebo-induced changes in brain activity or connectivity directly overlap with the brain and brain–body circuits implicated in mental disorders and broader health states across medicine (eg, pain, mood, and fatigue). Therefore, for what was once considered implausible, this overlap offers a tangible neurobiological mechanism for how placebo effects can legitimately improve relevant symptoms.”
III. Approaches for applying placebo effects in clinical practice
“Compared with typical treatment options, placebos can have fewer side-effects and reduced costs while sometimes maintaining similar efficacy. Given these potential benefits, a natural question arises: can, or should, placebos or components underlying placebo effects be harnessed in clinical practice?”
They review four types of placebos.
Deceptive placebos
“Deceptive placebos are those given to patients without telling them that the treatment is inert, and can either be pure or impure depending on their composition. The placebos most common in clinical trials are typically pure and considered completely inert, such as pills containing microcrystalline cellulose… Concealed pure placebos are regularly used in clinical trials with informed consent.”
Impure placebos
“By contrast, impure placebos have active ingredients but do not have established evidence for treating a given condition. Examples of common impure placebos are nutraceuticals, vitamins, or low-dose medications that have not been shown to have specific benefits for the target condition. Active brain stimulation that is perceptible but at an intensity, frequency, or location outside of the clinically relevant parameters could also be considered an impure placebo.
“To our knowledge, impure placebo has never been formally discussed in codes of ethics, but deceptive administration of impure placebos might similarly violate ethical principles of patient autonomy and informed consent. Nevertheless, use of impure placebos is common, and many practitioners consider it permissible. In one survey, 97% of physicians in the UK reported giving impure placebos at least once in their career, and 77% reported giving impure placebos at least weekly.”
Authorised deception
“A more ethical alternative to deceptive placebos is authorised deception, in which the patient is informed that some information will be withheld without specifying exactly what the deception is. In research contexts, the researcher asks the participant whether they agree with this deception during the consent procedure; the study only proceeds if they consent. Knowing about this potential deception does not necessarily weaken placebo effects. In one study, for example, participants received a placebo cream to reduce pain. One group were told that some part of the procedure involved deception, while the other group were not. The two groups showed no differences in the magnitude of their placebo effects or in their consent rates.”
Open-label placebos
“More transparent than authorised deception, open-label placebos are given with the patient having full knowledge that they are receiving placebo. Clinicians in open-label placebo studies typically give patients additional information for context: placebos can be powerful; the body can automatically respond to placebos in a clinical situation; placebos change neurotransmitters and brain activations; maintaining a positive attitude is helpful but not necessary for the treatment to work; and closely following the treatment regimen is still important…
“Open-label placebos have shown benefits across a range of disorders, including chronic pain, major depressive disorder, cancer-related fatigue, and irritable bowel syndrome. A 2023 meta-analysis found that open-label placebos led to stronger improvements than no-treatment or wait-list controls… Few long-term, open-label placebo studies exist and the current evidence is mixed. Three major RCTs for chronic lower back pain all showed positive results compared with the no-treatment group for up to 4 weeks.”
Optimising contextual factors in non-placebo treatments
“Perhaps the least controversial way to apply placebo research in the clinic is to harness the underlying principles of placebo effects rather than directly prescribe any kind of placebo. A range of situational factors contribute to placebo effects, including the clinician’s behaviour, patient–clinician relationship, perceived intensiveness of the treatment, treatment environment, and broader cultural factors. For example, the specific suggestions that clinicians provide and whether the clinician appears compassionate and competent contribute to treatment effects. Carefully controlling and optimising these factors could potentially increase the overall effectiveness of non-placebo treatments or therapeutic interactions.”
They note research barriers, including “applying such situational factors is that there is little research on how manipulating placebo effects in study contexts might generalise to non-placebo treatments in the clinic.” They continue: “If a compassionate and competent clinician could produce strong placebo effects, these effects might similarly increase the effectiveness of other interventions. A recent survey found that 164 (79%) of 207 physicians believed enhancing situational factors that contribute to placebo effects might be a way of improving paediatric patient care for non-placebo treatments.”
A few thoughts:
1. This is an excellent paper on a fascinating topic.
2. The summary above misses some of the richness and details of the paper itself.
3. The authors focus on depression. “A well-designed prospective placebo-focused depression RCT showed that pre-treatment patient expectancies are a substantial mediator of therapeutic responses. In this variant of an open-hidden paradigm, if participants were told they would be receiving placebo but actually received citalopram, their Hamilton Depression Rating Scale scores were significantly worse than participants who were told they had 100% chance of receiving citalopram.”
4. The clinical tips are good, including “overlapping psychotherapy sessions.”
5. H/t Dr. Ari Zaretsky.
The full Lancet Psychiatry paper can be found here:
https://www.thelancet.com/journals/lanpsy/article/PIIS2215-0366(25)00340-2/abstract
Selection 2: “One-Year Mortality Among Opioid Overdose Survivors”
Robert A. Kleinman, Sarah Larney, Naheemot Olaoluwa Sule, et al.
JAMA, 7 May 2026 Online First
Previous studies estimated 1-year mortality after emergency department visits for nonfatal opioid overdoses at approximately 5.5%. However, those studies were conducted before the widespread availability of fentanyl.
Here’s what they did:
- They drew on administrative health data from Ontario, Canada (population 16.1 million).
- “Individuals aged 15 to 105 years were included if they had an emergency department visit for a nonfatal opioid overdose between July 1, 2017, and June 30, 2023, and were discharged from the emergency department.”
- The primary outcome: “The primary outcome was death and the secondary outcome was opioid overdose, defined as a fatal opioid overdose or an emergency department visit for a nonfatal opioid overdose”
Here’s what they found:
- The cohort included 28 488 individuals with visits to 179 emergency departments.
- Demographics. Most were male (65.9%) and the mean age was 38.9 years.
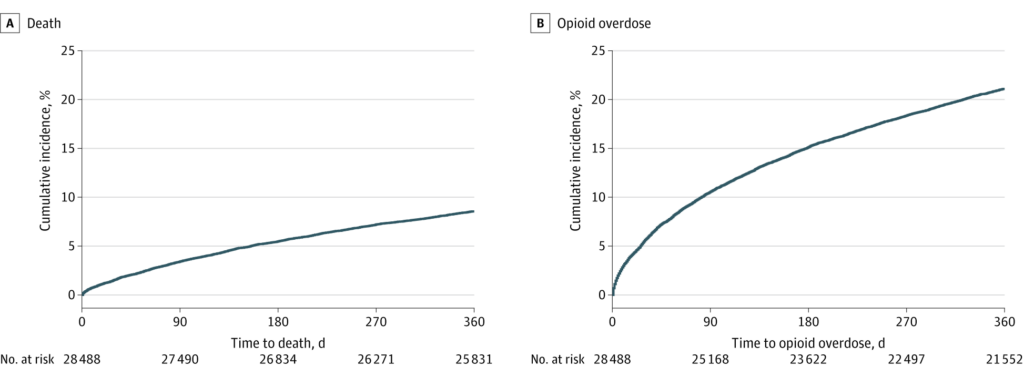
- Outcomes. “Within 1 year, 8.6% of individuals died (91.4 per 1000 person-years…), with 63.7% of deaths from opioid overdoses. During the 1-year follow-up, 21.2% of individuals had at least 1 repeat opioid overdose (252.5 per 1000 person-years…). One-year mortality was particularly elevated among individuals with a prior opioid overdose in the preceding 5 years (11.5%; 123.6 per 1000 person-years…).”
- Timing. “Incidence rates were highest immediately after discharge, although deaths and opioid overdoses continued throughout follow-up. Within 7 days of discharge, 0.6% of individuals died (323.2 per 1000 person-years…) and 2.4% of individuals experienced a repeat opioid overdose (1279.0 per 1000 person-years…). Within 30 days, 1.6% of individuals died (190.5 per 1000 person-years…) and 5.6% experienced a repeat opioid overdose (710.9 per 1000 person-years…).”
A few thoughts:
1. This is an excellent research letter, shedding light on an important problem with solid data.
2. The main finding: “In this population-based cohort, 1-year mortality after an emergency department visit for an opioid overdose was 8.6%, and 21.2% of individuals experienced a repeat opioid overdose.” The percentage was much higher than in previous studies; fentanyl is a game-changer.
3. The authors see practical implications: “This study identified high risks of death and repeat opioid overdose among survivors of nonfatal opioid overdose in the fentanyl era, with particularly elevated risks in the days immediately following emergency department discharge, underscoring the need for tailored clinical interventions for this high-risk population.”
4. Like all studies, there are limitations. The authors note several, including: “The study focused on individuals discharged from emergency departments and did not include individuals admitted to the hospital.”
The full JAMA research letter can be found here:
https://jamanetwork.com/journals/jama/article-abstract/2848748
Selection 3: “Mental health awareness campaigns can have unintended consequences”
Lester Liao, Elia Abi-Jaoude, and Michael Inzlicht
The Globe and Mail, 6 April 2026

A 10-year-old girl watches YouTube with a friend during their after-school program. An ad pops up. A two-minute quiz can tell her if she has ADHD. She takes it, of course. And just like that, she comes home to talk to her child psychiatrist dad about how she has ADHD.
This story is not unique. Mental health awareness has become one of Canada’s most visible public health projects. The messaging is everywhere: Bell Let’s Talk, school wellness e-mails, workplace campaigns, social media initiatives.
Yet despite these efforts, population-level mental health keeps declining. Medication use is increasing. How can this be?
So begins an essay by Liao et al.
They focus on mental health awareness campaigns. “While increased awareness of mental-health issues means we are surely detecting some of what was previously overlooked, drawing attention to the very problems we are trying to reduce may also magnify them… Encountering certain ideas can influence how we see ourselves and can change our behaviour. In fact, just telling people about ADHD can make them think that they have it, even when they have reported no significant symptoms beforehand. Schoolwide interventions for anxiety and depression can exacerbate symptoms in the short term. And copycat suicide is real.”
They feel that awareness can be problematic. “[Awareness efforts] frame ordinary experiences as medical problems. While destigmatization is the goal, medicalization becomes the norm. Anxiety over a test becomes not a passing experience but a symptom of a larger disorder. Sadness becomes depression. Social discomforts are reconceived as ‘autistic traits.’
“The trouble with increasing diagnoses is that medications for what’s identified as a medical problem become a logical next step. We reach for pharmaceutical solutions when social or behavioural approaches might serve us better – despite the fact that medicines can carry real side effects… Nausea from chemotherapy may be an acceptable trade-off. But appetite and growth suppression from psychostimulants in a child with borderline attention difficulties is more concerning. Perhaps we ought to ask more questions about our screen-riddled, sedentary culture.”
They note real-world consequences. “When attention and resources are directed to broader populations, those with the most debilitating illnesses are the true victims. Wait-lists for services become interminable. Conversation becomes dominated by those who are the loudest and most functional, not those who are so affected they cannot even speak. This leads to popular images portraying disorders such as autism as far less impairing than they really are, which only further marginalizes the greatest sufferers.”
They call for more research. “Some people do benefit from campaigns, and research could explore further how to reach the few with the most needs without ensnaring the many.”
A few thoughts:
1. This is a well-written essay.
2. The topic is relevant. Consider: during my last shift in the ED, I saw a patient who was convinced that her shyness was autism and a university student who confused the boredom of his required math course with the diagnosis of ADHD.
3. The authors focus on mental health campaigns, like Canada’s Bell Let’s Talk. Is that a bit simplistic? There are also paid advertisements from drug companies and social media posts from non-experts (and we can all agree that a well-meaning TikTok video isn’t a substitute for the clinical judgment of a clinician). The former suggests the need for better regulations and the latter could be addressed with social media content created by experts.
The full Globe and Mail essay can be found here:
https://www.theglobeandmail.com/opinion/article-mental-health-awareness-campaigns-consequences
Reading of the Week. Every week I pick articles and papers from the world of Psychiatry.
May 27, 2026 at 4:04 pm
I really enjoyed reading about these articles as a substitute for your in-person educational rounds. What stood out to me is the tension between increasing mental health awareness and over-medicalizing normal human distress. The placebo discussion reinforces how powerful the therapeutic relationship and hope can be in healing and not just medication alone. Thanks for sharing!