From the Editor
He occasionally used cannabis in his 20s and did not try crystal methamphetamine until after his 35th birthday. He readily admits to problematic use, undermining his relationship and leading to the loss of his job. In my office, he describes his desperation and asks: Is there a medication that could help?
In the first selection, a new paper published in JAMA Psychiatry, Rebecca McKetin (of the University of New South Wales) and her co-authors attempt to answer that question. In an impressive new study, they report the results of a phase 3, parallel-group, double-blind, placebo-controlled RCT involving 344 people with moderate or severe methamphetamine use disorder and a trial of mirtazapine or placebo. “The results of this randomized clinical trial confirm that mirtazapine can be used in routine clinical practice to facilitate a reduction in methamphetamine use among people with a moderate to severe methamphetamine use disorder.” We consider the paper and its implications.

In the second selection, Shaddy K. Saba and Dr. William B. Weeks (both of New York University) write on AI and patients. In a commentary for JAMA Psychiatry, the authors observe that an increasing number of patients are turning to AI and argue that clinicians need to become actively involved. “The question is not whether AI is good or bad for mental health: like many other patient behaviors, it can be both and depends on context. The question for clinicians is whether to help patients navigate its use.”
Finally, in the third selection from Schizophrenia Bulletin, Oliver Delgaram-Nejad – who has schizophrenia – discusses his decision to stop medication. Reviewing his episodes of psychosis, he argues that antipsychotics are too problematic for him. “I have weighed the benefits and risks, and navigated the rather painful tension, of remaining on an antipsychotic and living with the side effects versus accepting the risk of relapse and reclaiming my energy.”
DG
Selection 1: “Mirtazapine for Methamphetamine Use Disorder: A Randomized Clinical Trial”
Rebecca McKetin, Steven Shoptaw, Lucy Saunders, et al.
JAMA Psychiatry, 1 April 2026 Online First

Methamphetamine is a highly addictive synthetic stimulant drug that is a growing global public health concern. An estimated 7.4 million people worldwide have a methamphetamine use disorder, which is associated with an elevated risk of psychosis, cardiovascular events, accidental injuries, suicide, homicide, and suboptimal neonatal outcomes. In the US, methamphetamine use is a leading cause of drug-related death… No medications are approved by the US Food and Drug Administration for methamphetamine use disorder, and only a few options have provided hope.The generic tetracyclic antidepressant mirtazapine is a promising candidate. Mirtazapine modulates dopamine function via its affinity for serotonin (5-hydroxytryptamine [5-HT]) 5-HT2A, 5-HT2C, and 5-HT3 receptors, and this is thought to mediate its ability to reduce methamphetamine’s effects and ameliorate the dopaminergic dysregulation seen in methamphetamine addiction. Mirtazapine also blocks central presynaptic α2-adrenergic receptors, which are dysregulated in methamphetamine addiction… Evidence supporting the use of mirtazapine for methamphetamine use derives from 2 single-site phase 2 trials, the first with 60 participants and the second with 120. Together, these studies showed a 14% difference in active vs placebo condition in methamphetamine-positive urine tests.
So begins a paper by McKetin et al.
Here’s what they did:
- They conducted a “phase 3, parallel-group, double-blind, placebo-controlled randomized clinical trial.”
- Six outpatient alcohol and other drug clinics in Australia were involved; the trial was done between November 16, 2022, and May 1, 2025.
- “Participants were randomized 1:1 to receive either mirtazapine (30 mg/day for 12 weeks) or a matching placebo tablet.”
- Inclusion criteria included that people “were aged 18 to 65 years, had a moderate or severe methamphetamine use disorder in the past year according to the DSM-5, had used methamphetamine at least twice weekly in the past 4 weeks Mirtazapine (30 mg daily for 12 weeks) or equivalent placebo…”
- Primary outcome: “the change in days of methamphetamine use in the past 28 days from baseline to week 12.”
Here’s what they found:
- 344 participants were randomized with 339 participants received the intervention (167 in the placebo group and 172 in the mirtazapine group).
- Demographics and use. The mean age was 42.0 years; 37.2% were female. Participants used methamphetamine for a median of 24 days of the previous 28 days at baseline.
- Reduction. “The mean reduction in days of methamphetamine use from baseline to week 12 was greater in the mirtazapine group (7.0 days of 28 days) than in the placebo group (4.8 days of 28 days; mean difference, 2.2 days…).”
- Side effects. “More participants in the mirtazapine group reported drowsiness (47% vs 33%) and weight gain (10% vs 3%).
- Discontinuation. “Forty participants (23%) discontinued mirtazapine due to adverse events compared to 25 participants (15%) in the placebo group.”
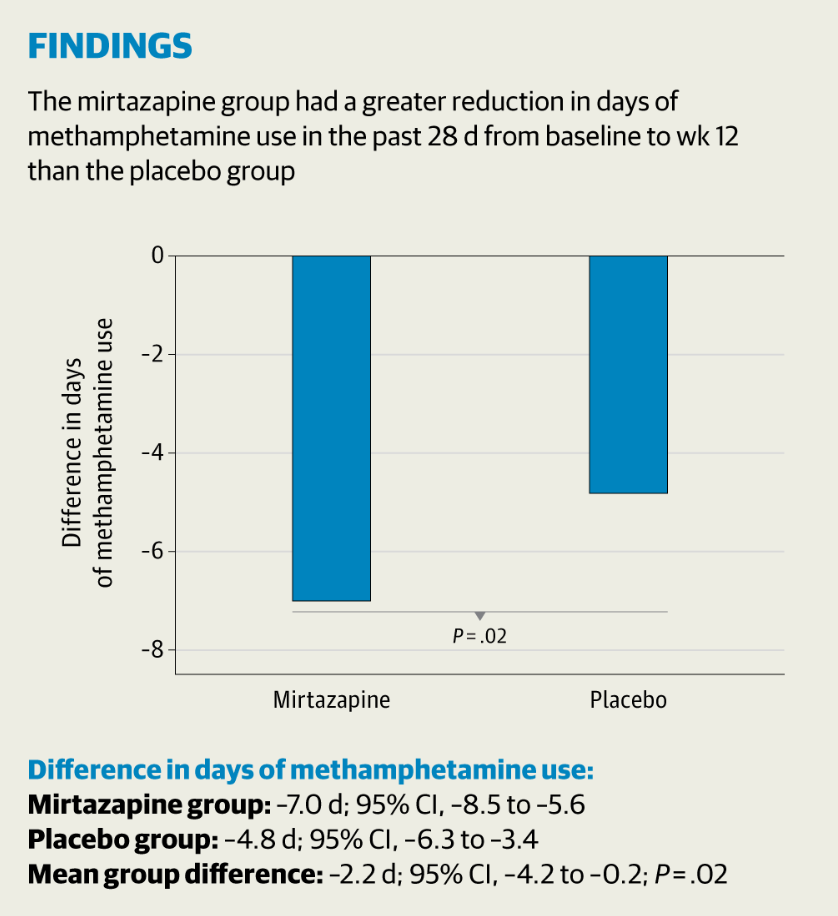
A few thoughts:
1. This is a good study, published in a major journal, with much to like – a practical question, a double-blinded design, a low attrition rate, and the inclusion of subpopulations not usually included in such work (such as those with depressive symptoms).
2. The main finding in a sentence: “In this randomized clinical trial, people with methamphetamine use disorder had a mean reduction of 7 days (out of 28 days) after 12 weeks of mirtazapine (30 mg/day) treatment, which was approximately 2 days more than the reduction seen with placebo.”
3. Needless to say, the authors see clear clinical implications. “The clinical significance of these findings lies in identifying a safe and cheap generic medication that can be prescribed to help people reduce methamphetamine use. The well-established safety profile of mirtazapine means it can be easily and safely prescribed in an outpatient setting with limited clinical oversight.”
4. The authors’ comment seems appropriate. Two days is helpful, though hardly miraculous. Still, mirtazapine use doesn’t have much of a downside.
5. Like all studies, there are limitations. The authors note several, including: the use of self-reporting of substance use.
The full JAMA Psychiatry paper can be found here:
https://jamanetwork.com/journals/jamapsychiatry/fullarticle/2847070
Selection 2: “Patients Use AI—Clinicians Should Ask How”
Shaddy K. Saba and William B. Weeks
JAMA Psychiatry, 1 April 2026 Online First

US individuals are turning to generative artificial intelligence (AI) as a mental health resource. More than 5 million US youth (13.1%) have sought mental health advice from AI, with rates reaching 22.2% among 18- to 21-year-olds. A 2025 study of adults with mental health conditions who use large language models reported nearly half use them for support, including for anxiety, depression, and personal advice. Users seek emotional support, companionship, psychoeducation, and help processing difficult experiences, likely between sessions and sometimes instead of clinical care altogether. While professional guidance focuses on how clinicians should use AI tools, conversations with patients about their AI use receive less attention.
So begins a paper by Saba and Weeks.
They focus on three areas.
Better understanding of the patient
“AI use offers a window into patients’ inner worlds: people likely turn to AI with concerns they might not share with clinicians, such as embarrassing fears, thoughts they worry will be judged, questions with seemingly obvious answers, and half-baked ideas. Encouraging patients to talk about AI use may prompt conversations about psychological experiences that would otherwise remain unexplored.”
Better understanding of their experiences
“AI shapes how patients process their experiences. A recent review of large language models in mental health contexts reported they can be overly sycophantic, offering validation without appropriate challenge; prone to hallucination, presenting incorrect or harmful information; and nondirective in ways that limit their therapeutic utility. Some users report responses can be too generic to apply to their actual circumstances. If clinicians do not know what their patients take from these tools, they cannot work with this content therapeutically.”
Better understanding of variables
“AI use parallels other routinely assessed clinical variables. Clinicians are curious about exercise, substance use, meditation applications, self-help books, and support group attendance precisely because understanding how patients cope is essential to helping them. As both a technology and a help-seeking behavior, AI use belongs in this category.”
They argue that: “Clinicians need not become AI experts, but basic literacy is necessary.” They add that they should understand the limitations of AI. “Clinicians must understand patient risks: while outputs sound authoritative, responses are often incomplete and can vary, and systems lack crisis protocols and accountability. Research on AI use in mental health is nascent and challenging to conduct. More concerning, AI may provide harmful information, fail to appropriately respond to suicidal ideation, reinforce harmful behaviors, or reflect social stigma and biases, including against racial and ethnic minority groups and individuals with serious mental illness.”
How to speak about AI. They offer several suggestions:
- “Normalizing AI use for mental health and asking questions with openness signals curiosity, and patients are more likely to share when they do not detect disapproval.”
- “Exploring benefits before concerns demonstrates genuine interest and helps clinicians understand the specific role AI is playing in patients’ lives.”
- “Asking about patients’ concerns before sharing clinicians’ concerns opens the conversation about harms.”
“Without routine assessment, patients are relating to these tools in ways clinicians cannot observe, developing habits they cannot shape, and potentially encountering harms they cannot prevent.”
A few thoughts:
1. The authors are practical.
2. The problem set is very new. But the approach – listening to patients, cautioning them about risks – isn’t.
3. Is AI good or bad for mental health? I like the authors’ comment: “like many other patient behaviors, it can be both and depends on context.”
4. AI and patient care have been considered in past readings. In August, we looked at a JAMA paper by Dr. Kumara Raja Sundar (of Kaiser Permanente Washington) who commented on patients using ChatGPT. The author, a family doctor, noted that many physicians can be paternalistic – but he urged against that instinct. “If patients are arming themselves with information to be heard, our task as clinicians is to meet them with recognition, not resistance.” That Reading can be found here:
The full JAMA Psychiatry commentary can be found here:
https://jamanetwork.com/journals/jamapsychiatry/article-abstract/2847068
Selection 2: “When Preston Begins to Feel Again: Discontinuation of Maintenance Antipsychotic Therapy”
Oliver Delgaram-Nejad
Schizophrenia Bulletin, 11 December 2026 Online First

This piece is concerned with my recent decision to discontinue maintenance antipsychotic therapy, after having been on amisulpride (200 mg daily) for around 3 years. I should start by saying that I have always been a vocal proponent of maintenance antipsychotics. Psychosis is not a pleasant experience, and for many it is even dangerous. I recognize that there will be some people for whom lifelong medication is sensible if not necessary. I want to, however, outline here some reasons for why discontinuation may be appropriate in some cases.
So begins an essay by Delgaram-Nejad.
He describes his experiences with psychosis. “I was hospitalized in 2014 for around 6 weeks. Due to a diagnosis of Wolff-Parkinson-White syndrome, I spent most of that time unmedicated. After spending a few days on 25 mg of quetiapine, my symptoms abruptly resolved. It is possible that I am a super responder to antipsychotic medication, but a part of me suspects that my episode may have run a natural course. Now, I am not suggesting that acutely psychotic patients should be left unmedicated in the expectation that this should happen for everyone, not least because of what the literature tells us about duration of untreated psychosis and poorer prognosis. I do think it is noteworthy, though, that I experienced such a rapid recovery on such a low dose of medication administered over such a short time frame. It raises the question, at least in my own case, of whether psychosis can resolve on its own in at least some cases.”
He struggles with the side effects of the quetiapine with brain fog and sedation. “We explored aripiprazole as an alternative given its liability to increase energy levels in some people. I spent around a week on the new medication before noticing signs of relapse and asking to switch back to quetiapine. I also developed neuroleptic malignant syndrome, and so we agreed to try discontinuation. I remained relapse free, and unmedicated, for the following 8 years.” He did relapse and recovered on amisulpride.
He wonders if he really needs antipsychotic medications. “Throughout all of this, I suspected that the antipsychotic may have been partly responsible for my tiredness and fatigue, which despite medical investigation had no established physical cause. I had long said that I did not want to stop taking my antipsychotic, as I was deeply fearful of a relapse. In recent months, however, I have started to question whether discontinuing my medication could be what’s needed to regain employment. My role in the intensive care unit was essentially impossible to sustain while feeling constantly exhausted, without significant motivation, and while struggling with oversleeping. These same side effects have also rendered me a rather lazy father, about which I feel very guilty.”
Does he have a spiritual problem? “I have begun to wonder whether this sense of loss is fundamentally a spiritual problem, rather than a neurochemical or psychogenic one. Although I trained first as a literary analyst and later a social scientist, I have recently found myself drawn to spiritual topics and questions. It is probably no surprise that this is coinciding with discontinuation of antipsychotic medication. Whereas I would previously have found this perilous or frightening, I am now embracing my tendencies toward overinclusive thinking, cluster A traits, and related symptoms as spiritual gifts, rather than symptoms of pathology. Now, I recognize that this may not be wise, or particularly logically grounded, but I have nevertheless noticed a significant improvement in my mood, motivation, and quality of life.”
“In all honesty, I did not expect myself to be taking this particular position. I have however found that addressing these issues has resulted in a renewed sense of purpose, confidence, and clarity.”
He closes on an ambitious note. “I have moved from the perspective of feeling unable to return to work long-term to formulating a clear plan of change.”
A few thoughts:
1. This is a thoughtful and well written essay.
2. We may disagree with his conclusion, but he isn’t rash or impulsive.
3. Who doesn’t admire his goal?
The full Schizophrenia Bulletin paper can be found here:
https://academic.oup.com/schizophreniabulletin/advance-article/doi/10.1093/schbul/sbaf226/8377360
Reading of the Week. Every week I pick articles and papers from the world of Psychiatry.
Recent Comments