From the Editor
Some patients with depression don’t show improvement. Among the reasons why: they may stop treatment too early. For decades, research on improving outcomes has included biomarkers, trying to better pair illness with treatment. But what about following patient preferences in care decisions? Can this help bolster medication compliance and thus improve outcomes?
In a new, compelling paper that was just published in JAMA, Dr. Andrea Cipriani (of the University of Oxford) and his co-authors attempt to answer these questions. They describe a study involving 540 participants in 47 sites. Participants were randomized to receive either the usual care or care using the PETRUSHKA tool, “a web-based clinical decision-support system combining clinical and demographic predictors with patient preferences to personalize antidepressant treatment.” The results are impressive. “Compared with usual care, participants with MDD whose antidepressant was selected using the PETRUSHKA tool had a reduced risk by approximately 40% of discontinuing their antidepressant during the first 8 weeks of treatment.” We consider the paper and its implications, as well as the accompanying Editorial.

The impact of social media on children and adolescents is much discussed; recently, Australia banned youth who are under 16 years of age from using platforms like Instagram. In this week’s other selection, from JAMA Pediatrics, Ben Singh (of the University of South Australia) and his co-authors analyzed social media’s association with well-being. They describe a cohort study of more than 100 000 Australian adolescents who were followed for three years. They found: “a U-shaped association emerged where moderate social media use was associated with the best well-being outcomes, while both no use and highest use were associated with poorer well-being.”
Note: there will be no Readings for the next two weeks.
DG
Selection 1: “A Decision-Support System to Personalize Antidepressant Treatment in Major Depressive Disorder: A Randomized Clinical Trial”
Andrea Cipriani, Karen Barros Parron Fernandes, Benoit H. Mulsant, et al.
JAMA, 4 March 2026 Online First

With 280 million people currently affected in the world, major depressive disorder (MDD) is one of the leading causes of global health burden, and it is forecast to increase further by 2050. Antidepressants are recommended as one of the first-line options for people with moderate to severe symptoms; however, a key challenge is how to best implement these treatments in clinical practice. Prescription of antidepressants has increased over the last 25 years, but the majority of people with MDD take them for too short a period. The initial adverse events associated with antidepressant use contribute to the suboptimal duration of treatment, reducing the clinical effectiveness of these medications. This is exacerbated by the inability of clinicians to reliably predict which antidepressant will cause adverse events in each individual patient, and which medication will be the most effective for each patient…
In some fields of medicine, such as oncology and cardiology, prediction tools play an important role in clinical decision-making. However, in psychiatry, prediction of treatment outcomes is challenging, especially for patients with MDD, because treatment trajectories are influenced by the complex etiology and heterogeneous symptomatology of the disorder. Previous studies have found that sociodemographic features (eg, age, biological sex, marital status) and clinical characteristics (eg, symptom severity, prior treatments, comorbidities) are associated with individual treatment outcomes in MDD. However, there is no consensus on which pretreatment characteristics can accurately predict efficacy and acceptability of specific antidepressants. Moreover, the acceptance of adverse events by patients is heterogeneous and some patients may be more willing to tolerate certain adverse events over others based on their personal circumstances.
So begins a paper by Cipriani et al.
Here’s what they did:
- They conducted “an international, multicenter, superiority randomized clinical trial conducted at 47 sites in both primary care (in Brazil and in the UK) and secondary care (in Canada).”
- Eligible patients had a diagnosis of MDD and were between the ages of 18 and 74.
- Exclusion criteria included treatment-resistant depression.
- There were 47 sites in three countries (Brazil, Canada, and the UK).
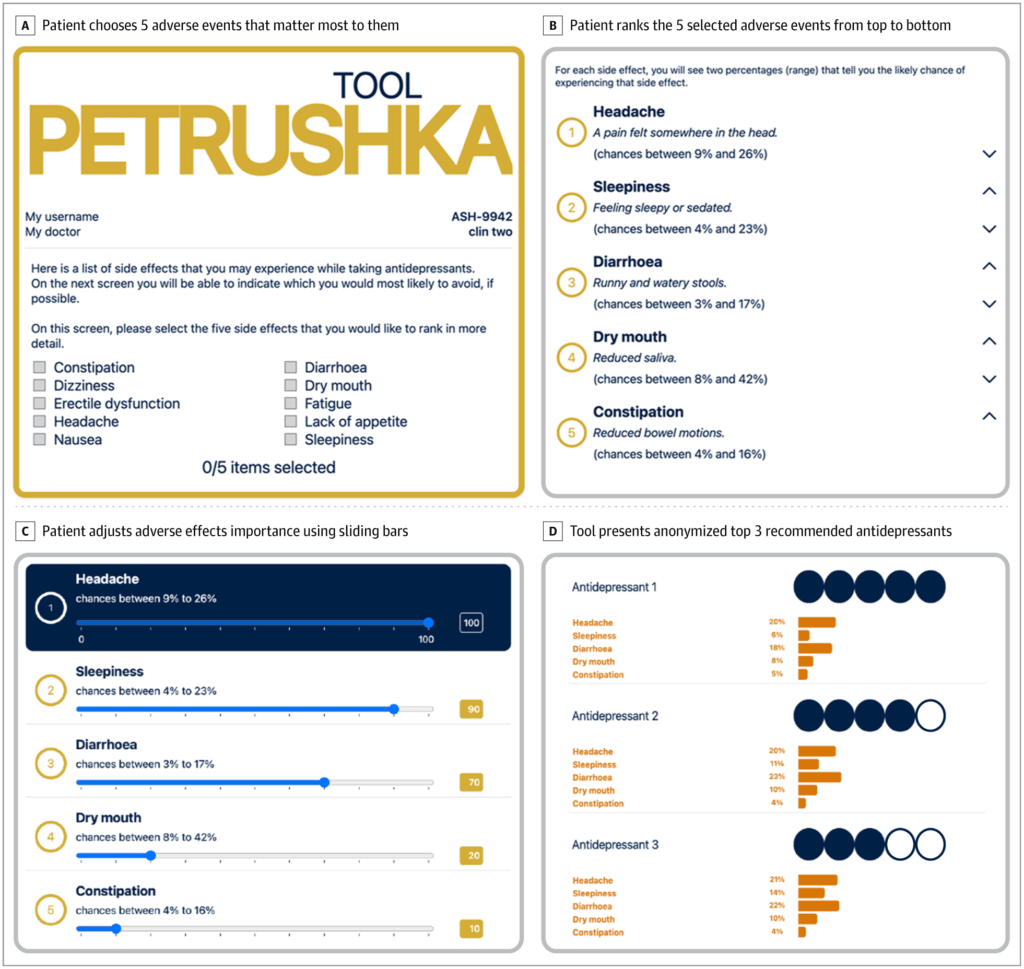
- Participants were randomized to a clinical decision-support system or to the usual care. “The PETRUSHKA tool was used to select the antidepressant with the best profile for each individual. Based on the participant’s individual demographic and clinical characteristics, the PETRUSHKA tool predicted the performance of each drug for each outcome. These predictions were then combined with the patient’s preferences about avoiding particular adverse events to create a short list of the 3 best antidepressants…”
- Primary outcome: treatment discontinuation due to any cause at 8 weeks.
Here’s what they found:
- A total of 540 participants were randomized to an evidence-based clinical decision-support system (271) or usual care (269).
- Demographics & illness experience. Most participants were female (58%) with a median age of 35 years. The PHQ-9 mean score was 16.6; GAD-7 mean score, 11.5.
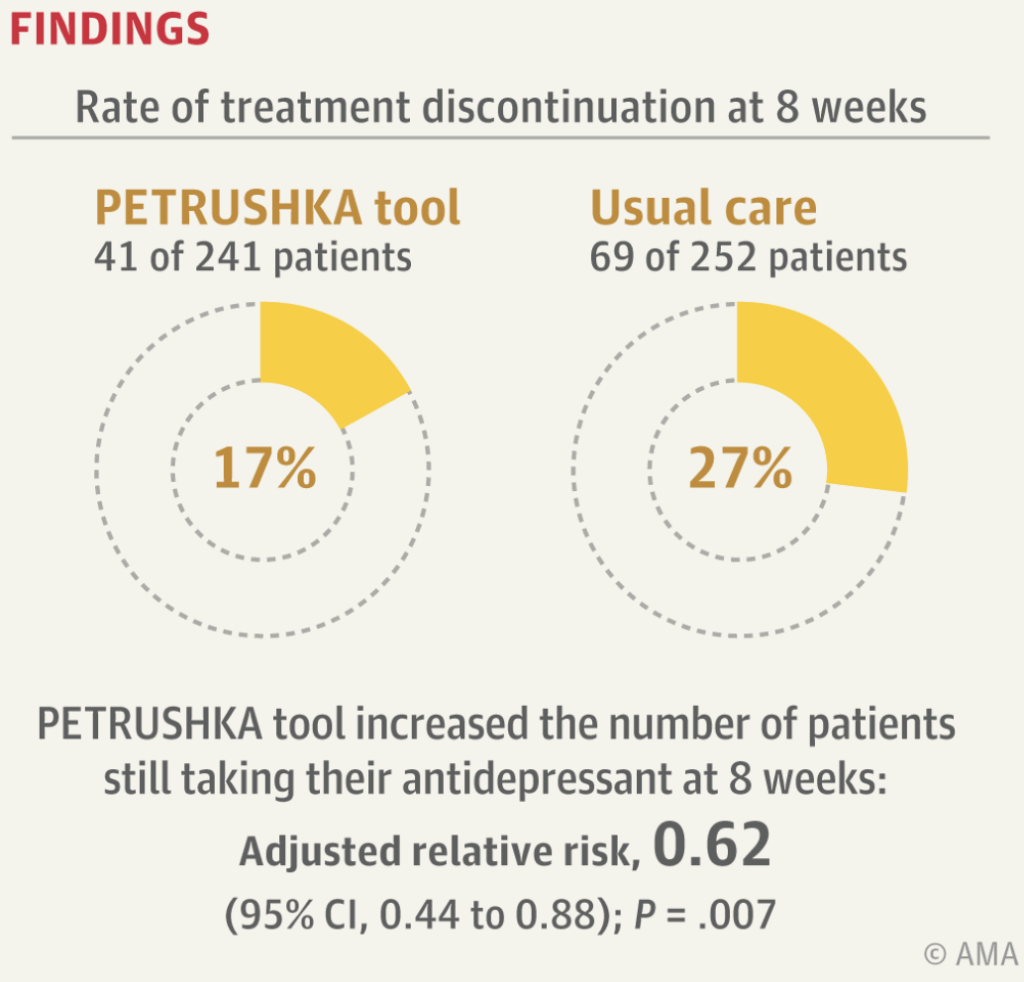
- At eight weeks. 17% in the PETRUSHKA group discontinued the prescribed antidepressant due to any cause vs 27% in the usual care group (adjusted relative risk, 0.62).
- Mood & anxiety. “For the assessment of depressive symptoms at 24 weeks, the mean PHQ-9 score was 7.1… in the PETRUSHKA group vs 9.2… in the usual care group (n = 129 in each group; adjusted between-group mean difference, −1….). For the assessment of anxiety symptoms at 24 weeks, the mean GAD-7 score was 4.6… in the PETRUSHKA group (n = 133) vs 5.8… in the usual care group (n = 126) (adjusted between-group mean difference, −1.39…).”
- Safety. “There were 2 serious adverse events in the PETRUSHKA group; neither was judged to be caused by the PETRUSHKA tool or the prescribed antidepressant.”
A few thoughts:
1. This is an important paper addressing a practical problem, published in a major journal.
2. The main finding in a sentence: “Compared with usual care, use of the PETRUSHKA tool increased the number of patients still taking their antidepressant at 8 weeks and improved depressive and anxiety symptoms at 24 weeks.” Nice.
3. It’s a solid result.
4. There is much to like about this study, including that it was conducted over many sites.
5. How exactly does the tool work? The paper gives details, describing how participants can rank adverse events and then have antidepressant choices ranked accordingly. See the figure below.
6. Not surprisingly, the tool tended to work better in primary care settings (Brazil and the UK) than in psychiatric ones (Canada). The authors see practical implications: “our results suggest that the use of the PETRUSHKA tool could be ideally placed in clinical settings where MDD is managed by other clinicians at community hospitals and specialist outpatient clinics (neurology and cardiology clinics or another specialty), in low-resource or rural areas, and in low- and middle-income countries.” The PETRUSHKA tool is clever and new – but is the core lesson not so new? That is, patient empowerment yields better outcomes.
7. Like all studies, there are limitations. The authors note several, including with the open design. “A double-blind trial would have been, in theory, the best study design to address our research question. However, it was not ethically possible to do so because the PETRUSHKA tool would have produced personalized recommendations, whereas the placebo tool would have produced random recommendations that could have harmed the participants.”
8. The paper appears with an Editorial by Dr. Greg Simon (of Kaiser Permanente Washington Health Research Institute). He writes: “Early research to personalize depression treatment yielded decades of disappointment.”

Greg Simon
He adds: “Subtyping depression based on clinical characteristics did not predict patient response to specific medications, reflecting both the heterogeneity of depression and the wide range of clinical and nonclinical factors affecting outcomes. Pharmacogenomic, imaging, and other biomarker research failed to identify reproducible predictors of specific medication responses…”
He finds much to like in this study. “The rigorous randomized comparison in the trial demonstrated significant improvements in the outcomes that are relevant to patients and clinicians.” And he sees larger lessons. “The PETRUSHKA trial… suggests some useful general principles for personalizing antidepressant treatment in the near term. Given the current disappointing state of knowledge regarding subtypes of depression and mechanisms of action in antidepressant medications, personalization based on differences in expected effectiveness and adverse events will likely benefit patients sooner than personalization based on differences in presumed therapeutic mechanism.”
He concludes on a practical note: “While clinicians wait for genomics, proteomics, or another field to reveal underlying biology and transform care for depression, patient preference appears to offer important clinical benefit with little risk, cost, or treatment delay.”
The Editorial can be found here:
https://jamanetwork.com/journals/jama/article-abstract/2845914
The full JAMA paper can be found here:
https://jamanetwork.com/journals/jama/article-abstract/2845910
Selection 2: “Social Media Use and Well-Being Across Adolescent Development”
Ben Singh, Mason Zhou, Rachel Curtis, et al.
JAMA Pediatrics, 12 January 2026 Online First

Despite social media’s central role in adolescent life, its implications for well-being are far from clear. On one hand, social media platforms support social connection, self-expression, and access to information; on the other, heavy use has been associated with emotional distress, social comparison, and cyberbullying… Much of the evidence addressing the psychological impacts of social media to date has been cross-sectional. Such studies consistently find that adolescents reporting higher social media use also report poorer outcomes, including depressive symptoms, anxiety, lower life satisfaction, and disturbed sleep. A 2022 umbrella review of 25 literature reviews (meta-analyses, systematic reviews, and narrative reviews) found most studies concluded that the associations between social media use and adolescent mental health outcomes were weak, while a small number of reviews found a more substantial association. However, the evidence base is overreliant on cross-sectional studies, which cannot reveal directionality: is social media driving poor well-being, or do young people who are struggling already turn to social media more?
Longitudinal studies provide a more cautious perspective. For example, in a 2-year cohort study of 594 adolescents (grades 6-8; mean age, approximately 12.2 years) in Ontario, Canada, researchers found that daily social media use did not predict subsequent depressive symptoms once baseline characteristics were accounted for, although higher depressive symptoms did forecast increased social media use over time among girls… These findings suggest a dynamic rather than unidirectional association, highlighting that social media use is unlikely to pose a uniform risk across all adolescents and may instead reflect or exacerbate preexisting vulnerabilities.
So begins a paper by Singh et al.
Here’s what they did:
- They conducted a cohort study that included Australian students in grades 4 through 12 (2019-2022).
- In terms of social media use, they focused on after-school usage that was self-reported and “classified into 3 categories: none (0 h/wk), moderate (>0 to <12.5 h/wk), and highest (≥12.5 h/wk).”
- “Well-being was assessed concurrently with social media use during the annual school-based survey in each year of data collection.”
- Primary outcome: “overall well-being, measured as the mean score across 8 validated domains (happiness, optimism, life satisfaction, worry, sadness, perseverance, emotional regulation, and cognitive engagement), dichotomized as high vs low (<3 on a scale of 1-5).”
Here’s what they found:
- There were 100 991 adolescents included, with 173 533 observations.
- Demographics. 49.9% observations from female participants with a mean age of 13.5 years.
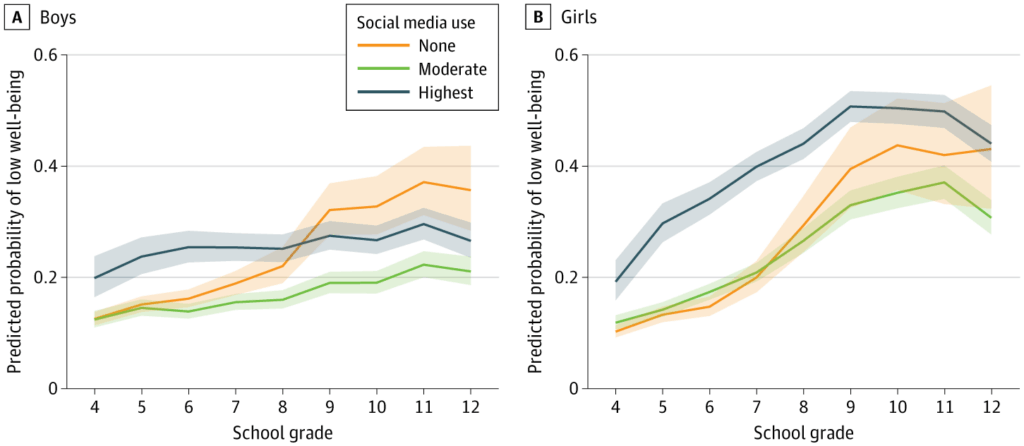
- Association. A U-shaped association was observed between after-school social media use and well-being. “Compared with moderate users, adolescents with the highest use had greater odds of low well-being (grades 7-9, girls: odds ratio [OR], 3.13; boys: OR, 2.25). Nonusers also had higher odds of low well-being in later adolescence (grades 10-12, girls: OR, 1.79; boys: OR, 3.00.” See figure below.
- Patterns. These patterns were consistent across survey years and robust to sensitivity analyses.
A few thoughts:
1. This is a good study, published in a major journal.
2. The core finding in a sentence: “Adolescents reporting moderate use generally demonstrated the most favorable well-being profiles, whereas both nonusers and those with the highest levels of use were at elevated risk for low well-being.”
3. The authors see clear practical implications: “Public health and school-based programs should promote balanced and constructive use of social media, integrating strategies that build digital resilience, emotional regulation, and critical appraisal of online content.”
4. The call for government action over social media has grown stronger in recent years. The study reminds us that the issue is nuanced and complicated.
5. A note of caution, though: this study looked at well-being – not depression or anxiety.
6. Like all studies, there are limitations. The authors note several, included the reliance of self-report data: “self-reported social media use may be prone to recall or social desirability bias; while responses were collected in a confidential format, participants were reidentifiable to enable linkage across survey years.”
The full JAMA Pediatrics paper can be found here:
https://jamanetwork.com/journals/jamapediatrics/article-abstract/2843720
Reading of the Week. Every week I pick articles and papers from the world of Psychiatry.
Recent Comments